Overview
Millions of people worldwide suffer pain and limited mobility as a result of damage to articular cartilage, most commonly in the knee and ankle. Unfortunately, unlike muscle or bone, damaged cartilage has a limited capacity for self-repair.
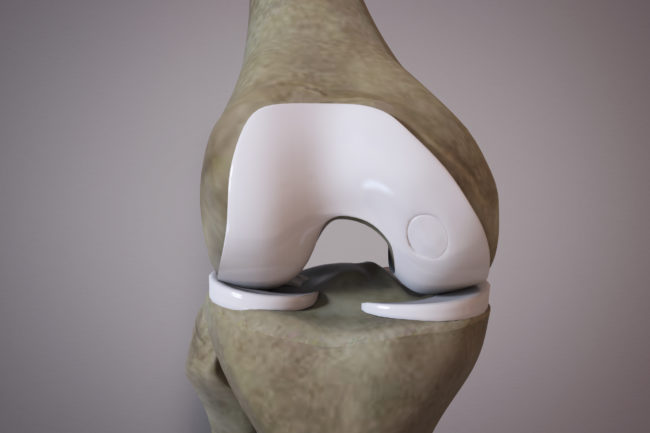
Orthocell’s Autologous Chondrocyte Implantation (OrthoACI™) offers treatment for symptomatic defects of the articulating cartilage of the joints. OrthoACI™ uses the patient’s own healthy cartilage cells called chondrocytes to assist the regeneration of damaged cartilage. OrthoACI™ is a highly customized treatment – each procedure is tailored to the individual patients’ requirements.
Product not available for sale in the EU/UK.
How it works
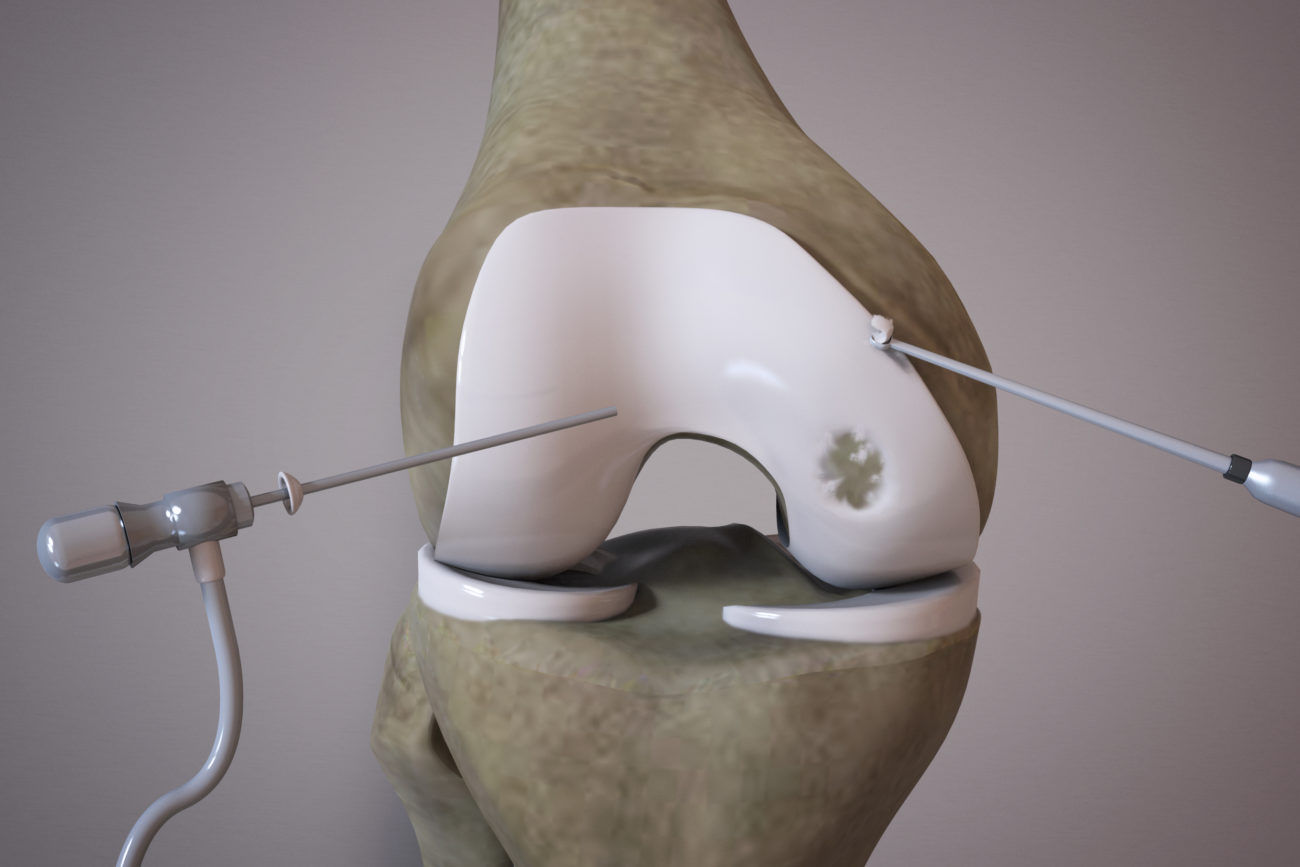
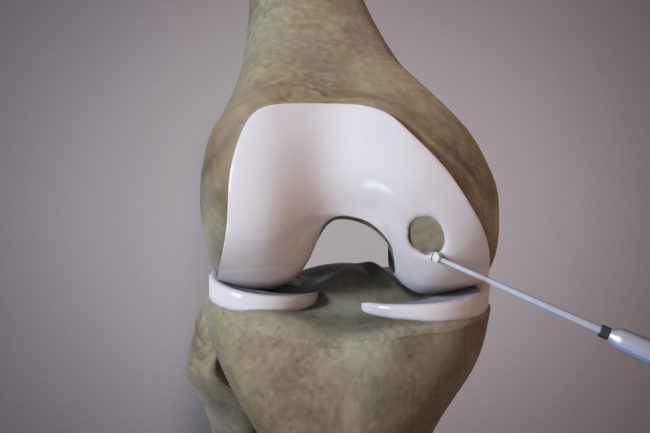
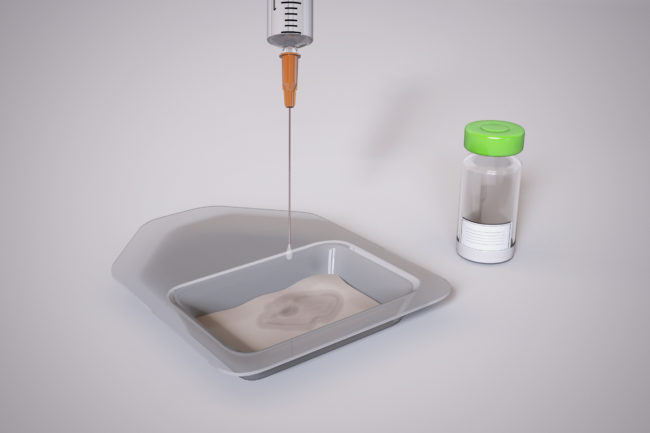
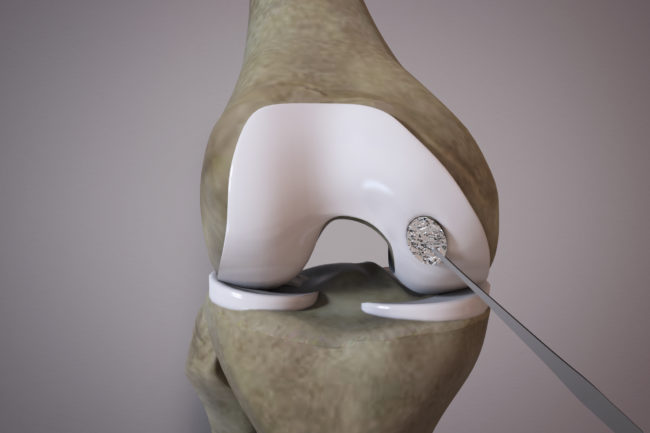
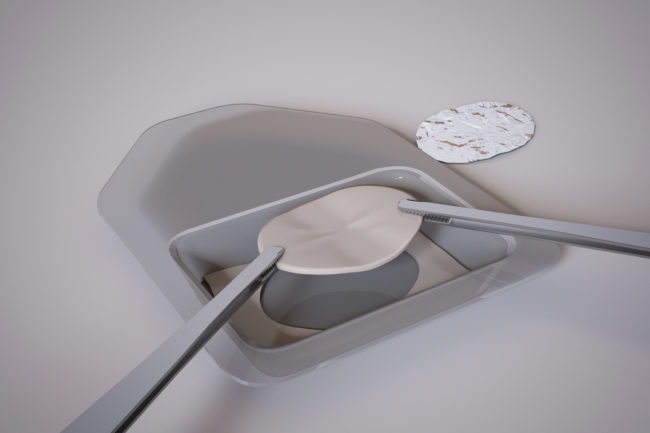
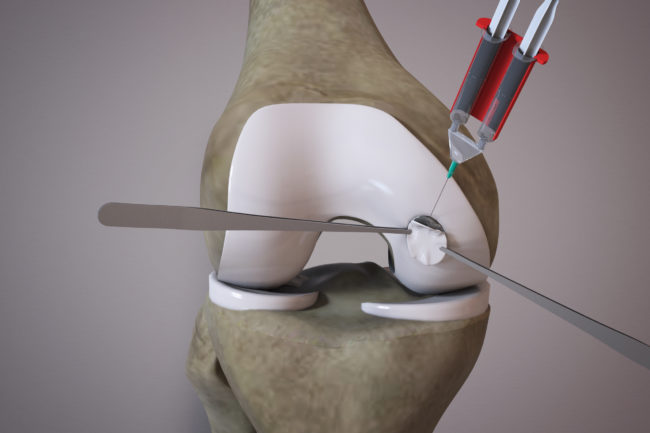
OrthoACI™ is a two-stage procedure. A biopsy of healthy articular cartilage using keyhole surgery is taken and sent to Orthocell’s quality-controlled laboratory where the chondrocytes are isolated and grown under controlled conditions for approximately 5 weeks. The cells are loaded on a custom-designed cell delivery scaffold which is implanted into the damaged joint by the orthopaedic surgeon through a small incision or keyhole surgery. The scaffold creates the optimal environment for the implant chondrocytes to begin regenerating new cartilage.
A rehabilitation program to slowly increase in weight bearing over a six-week period is essential to ensure the success of OrthoACI™. Return to sport and recreational activities should also be staged over the first six to nine months following OrthoACI™. This staged increase in intensity of activities protects the newly developing cartilage and allows cartilage maturation process to occur undisturbed.
Two-stage surgical technique
First stage
Webinars
Latest developments in the biological treatment of Cartilage
For more information
To find out more, get in touch via the contact form.